i do not really understand what the sheme are all about, pleas i whish to have some darectives about them.
So, what's your question? 
What?

Original post by epangaonanaherve
i do not really understand what the sheme are all about, pleas i whish to have some darectives about them.
What?

Well do you just want the answers or what don't you understand?
Do you know what ozonolysis followed by a reductive work up gives you? Do you know how to make an ester?
Have you heard of the Pinacol-Pinacollone rearrangement? Do you know how to remove carbonyl groups?
Do you know what ozonolysis followed by a reductive work up gives you? Do you know how to make an ester?
Have you heard of the Pinacol-Pinacollone rearrangement? Do you know how to remove carbonyl groups?
(edited 11 years ago)
Ozonolysis is a fairly simple mechanism you should have seen, if not it's easy to find on the internet.
The rearrangement is a shift of carbon group. Protonate one of the OH's to make a good leaving group, then attack down with the OH lone pair. The C-C orbital is of the correct alignment to overlap with the c-o sigma star, migrating across to form the spiro center.
The rearrangement is a shift of carbon group. Protonate one of the OH's to make a good leaving group, then attack down with the OH lone pair. The C-C orbital is of the correct alignment to overlap with the c-o sigma star, migrating across to form the spiro center.
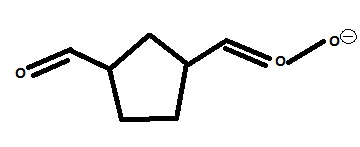
Sorry, I should have been more clear, I'm just clueless about part ii) for both questions and I think I have the mechanism for the first question wrong. I don't know what to do after I get:
Thanks for the help so far, I really appreciate it
Thanks for the help so far, I really appreciate it

Original post by ScaryKid_ScaringKids
Sorry, I should have been more clear, I'm just clueless about part ii) for both questions and I think I have the mechanism for the first question wrong. I don't know what to do after I get:

Thanks for the help so far, I really appreciate it
Thanks for the help so far, I really appreciate it

Usually I'd recommend diazomethane for conversion from an acid into a methyl ester, but in this case you also have a ketone so run the risk of CH2 insertion next to it. The safest way would be reaction with methanol and an acid catalyst. Diazomethane would probably be ok as the COOH proton is more reactive than the ketone C=O. Acid/alcohol esterification is an equilibrium so wouldn't give you an amazing yield, and the diazomethane method could have side reactions. I'm not sure which would give the best yield, but if I had to guess I'd reckon the diazomethane one would be better as long as you only use one equivalent.
As for part b) removal of carbonyl groups can be done by several methods.
The most tolerant for other functional groups is reaction with a di thiol and BF3 to form a thioacetal (the sulphur analogue to an acetal), which is then cleaved with Raney Nickel.
Another is the Wolff Kishner reduction. Reaction with hydrazine and hot conc OH-.
Lastly the clemmensen reduction. Zinc dissolving in conc HCl. But this can only really be used when the C=O is the only functional group present.
In this case any of the three above methods is fine.
(You could also reduce to the alcohol, tosylate then reaction with BH4- [a source of nucleophilic H] but this is more steps)
Lastly http://en.wikipedia.org/wiki/Ozonolysis#Reaction_mechanism
(edited 11 years ago)
I though oxidative workup would give you the straight carbonyls?
If you were to use SMe2 for the workup, you would definitely get the dicarbonyl.
If you were to use SMe2 for the workup, you would definitely get the dicarbonyl.
Original post by JMaydom
I though oxidative workup would give you the straight carbonyls?
If you were to use SMe2 for the workup, you would definitely get the dicarbonyl.
If you were to use SMe2 for the workup, you would definitely get the dicarbonyl.
Oxidative workup gives acid/ketone.
Me2S gives aldehyde/ketone
NaBH4 gives alcohol
Original post by illusionz
Usually I'd recommend diazomethane for conversion from an acid into a methyl ester, but in this case you also have a ketone so run the risk of CH2 insertion next to it. The safest way would be reaction with methanol and an acid catalyst. Diazomethane would probably be ok as the COOH proton is more reactive than the ketone C=O. Acid/alcohol esterification is an equilibrium so wouldn't give you an amazing yield, and the diazomethane method could have side reactions. I'm not sure which would give the best yield, but if I had to guess I'd reckon the diazomethane one would be better as long as you only use one equivalent.
As for part b) removal of carbonyl groups can be done by several methods.
The most tolerant for other functional groups is reaction with a di thiol and BF3 to form a thioacetal (the sulphur analogue to an acetal), which is then cleaved with Raney Nickel.
Another is the Wolff Kishner reduction. Reaction with hydrazine and hot conc OH-.
Lastly the clemmensen reduction. Zinc dissolving in conc HCl. But this can only really be used when the C=O is the only functional group present.
In this case any of the three above methods is fine.
(You could also reduce to the alcohol, tosylate then reaction with BH4- [a source of nucleophilic H] but this is more steps)
Lastly http://en.wikipedia.org/wiki/Ozonolysis#Reaction_mechanism
As for part b) removal of carbonyl groups can be done by several methods.
The most tolerant for other functional groups is reaction with a di thiol and BF3 to form a thioacetal (the sulphur analogue to an acetal), which is then cleaved with Raney Nickel.
Another is the Wolff Kishner reduction. Reaction with hydrazine and hot conc OH-.
Lastly the clemmensen reduction. Zinc dissolving in conc HCl. But this can only really be used when the C=O is the only functional group present.
In this case any of the three above methods is fine.
(You could also reduce to the alcohol, tosylate then reaction with BH4- [a source of nucleophilic H] but this is more steps)
Lastly http://en.wikipedia.org/wiki/Ozonolysis#Reaction_mechanism
As the resident diazo chemist, I feel reasonably confident in saying that the esterification would proceed cleanly. Of course, trimethylsilyldiazomethane in 7:30 toluene:methanol is safer, and works just as well.
Original post by EierVonSatan
So, what's your question? 
What?

What?

i can identify the structu
Original post by EierVonSatan
So, what's your question? 
What?

What?

i connot identify structure E,G,H and J
Original post by epangaonanaherve
i connot identify structure E,G,H and J
Their structures are given in the opening post?
You know that this is degree level, yes? Your profile says that you're 16...
Original post by EierVonSatan
Their structures are given in the opening post?
You know that this is degree level, yes? Your profile says that you're 16...
You know that this is degree level, yes? Your profile says that you're 16...
Background checks for the win!

Original post by cpchem
As the resident diazo chemist, I feel reasonably confident in saying that the esterification would proceed cleanly. Of course, trimethylsilyldiazomethane in 7:30 toluene:methanol is safer, and works just as well.
Oh yeah, of course.... :P
Good to know. Seemed logical that the deprotonation would be a faster reaction than insertion but have just been revising for a course which is all about selectivity in synthesis so that's always at the top of my mind!
Quick Reply
Related discussions
- Organic chemistry textbook (uni)
- aqa a level chemistry paper 2 assessed topics
- Chemistry at Uni?
- physical or inorganic??
- How can I become obsessed with chemistry ??
- Chemistry revision
- A level chemistry for a biology degree? (help please!!)
- Tips for physical A level chemistry OCR A?
- UCL Natural Sciences and streams
- Is Chemistry better at A-level than at GCSE?
- AS/A Level Chemistry Study Group 2023/2024
- How should I study organic chemistry?
- Biochem to Med chem
- Inorganic Chemistry (AQA) - A level
- Learning techniques
- Chemistry or Biochemistry?
- OCR A Chemistry Predictions
- Bath Mpharm Interview Questions
- I enjoy chem practicals more than bio practicals. Is a chem degree right for me?
- Organic, Physical or Inorganic Chemistry for tutoring?
Latest
Trending
Last reply 6 days ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 6 days ago
Im confused about this chemistry question, why does it form these products



