Cambridge Chemistry Challenge
Scroll to see replies
Original post by alow
Okay scratch that then. Groups/atoms in a molecule are said to be "chemically equivalent" or "in the same environment" if the structure of the molecule is such that they are joined to the same groups/atoms.
e.g.
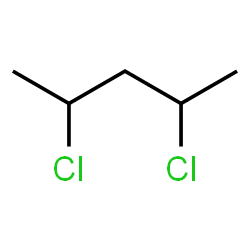
The molecule is symmetric about the 3rd carbon, so the protons on the 1st & 5th and 2nd & 4th are equivalent.
However the molecule need not be symmetric:

This molecule has almost no symmetry but there are equivalent proton environments on the phenyl substituent (the 6-memebered ring with alternating double bonds), as the 2/6 and 3/5 protons are equivalent (counting clockwise around the ring, starting where the phenyl is joined to the other part of the molecule).
There doesn't necessarily need to be symmetry: see above.
e.g.
The molecule is symmetric about the 3rd carbon, so the protons on the 1st & 5th and 2nd & 4th are equivalent.
However the molecule need not be symmetric:

This molecule has almost no symmetry but there are equivalent proton environments on the phenyl substituent (the 6-memebered ring with alternating double bonds), as the 2/6 and 3/5 protons are equivalent (counting clockwise around the ring, starting where the phenyl is joined to the other part of the molecule).
There doesn't necessarily need to be symmetry: see above.
Awesome this really helps, the environment thing has come up twice in vocabulary and it will really help with the questions.
Many thanks
I did this last year, I remember walking out very perplexed and thankful for how much easier my AS chemistry exams were. Ended up with a Silver which I was fairly happy with given that no work was put into it.
Posted from TSR Mobile
Posted from TSR Mobile
For the 2011 paper. why do the deuteriums have to be on the carbon not involved in the double bond rather than the double bonding carbon on the end of the propene, given that they're in the same environment?
(edited 7 years ago)
Original post by Klimenski
What do you mean by equivalent, the mark scheme shows 3 OH groups around the arsenic atom, how does environment link in with that?
where is the mark scheme?
Do you guys remeber your answers for the latest 2016 challenge sat today (23/06/16) or find a mock mark scheme?
Original post by Klimenski
Do you guys remeber your answers for the latest 2016 challenge sat today (23/06/16) or find a mock mark scheme?
the first question had something to do with extraction of titanium
Anyone remember answers to the 2016 paper?
Original post by krish10
Anyone remember answers to the 2016 paper?
got my marks back today!! I got a silver award!!! so chuffed!!
Hi all, I'm new to this thread and the Cambridge Chemistry Challenge, I've completed the first two June 2015 online challenge and i'm stuck on the Sliver Level Question which is titled 'Whose Nail?'. I'm a little bit confused as the image background shows the molecular model of C20H14N4 which is Porphin and the answer is not Porphin! So please help me with this question
Thank you all!!!

Thank you all!!!
Original post by Greenapplepear
Hi all, I'm new to this thread and the Cambridge Chemistry Challenge, I've completed the first two June 2015 online challenge and i'm stuck on the Sliver Level Question which is titled 'Whose Nail?'. I'm a little bit confused as the image background shows the molecular model of C20H14N4 which is Porphin and the answer is not Porphin! So please help me with this question
Thank you all!!!

Thank you all!!!
A picture would be useful.
Original post by alow
A picture would be useful.

Yes, here u go.
Thanks
Original post by Greenapplepear
Yes, here u go.
Thanks

Yes, here u go.
Thanks
Have fun.
https://www.wikiwand.com/hr/Willst%C3%A4tterov_%C4%8Davli%C4%87
Thank you so much!!!
Does the kelvin of any of the elements have to do with the answer
anyone here doing it in 2021?
Hello there, if anyone is still here doing the chemistry challenge puzzles, would someone be kind to share with me the hints to the roentgenium challenge in February 2015? I have no idea to what ion they expect us to use. I have done every single other chemistry challenges and this is my last one. Huge thanks.
Original post by cat19393
Hello there, if anyone is still here doing the chemistry challenge puzzles, would someone be kind to share with me the hints to the roentgenium challenge in February 2015? I have no idea to what ion they expect us to use. I have done every single other chemistry challenges and this is my last one. Huge thanks.
I got the answer (around 10^-6) using the two equations Ek = 1/2mv^2 and s = vt, equating them using v. You may also need to know from a level physics that V = WD / Q (voltage is work done per unit charge). To help you out the ion is the phenyl cation.
Original post by bigman_caramel
I got the answer (around 10^-6) using the two equations Ek = 1/2mv^2 and s = vt, equating them using v. You may also need to know from a level physics that V = WD / Q (voltage is work done per unit charge). To help you out the ion is the phenyl cation.
OMG THANK YOU SO MUCH!! Also may I please ask you for the silver level question for January 2015? I just need the question, not the answer. For some reason I can’t view the real question due to some glitch happening, instead I saw a wrong picture for another question on fireworks, which is wrong. thanks so much for helping me complete the challenges, this silver question is the real last one. 😊 (I know I said the Time of flight question is the last one in my post before but this is my real last one lol)
(edited 4 months ago)
Original post by cat19393
OMG THANK YOU SO MUCH!! Also may I please ask you for the silver level question for January 2015? I just need the question, not the answer. For some reason I can’t view the real question due to some glitch happening, instead I saw a wrong picture for another question on fireworks, which is wrong. thanks so much for helping me complete the challenges, this silver question is the real last one. 😊 (I know I said the Time of flight question is the last one in my post before but this is my real last one lol)
Idk sorry I get the fireworks image too lol
Original post by bigman_caramel
Idk sorry I get the fireworks image too lol
Oh, do you have the answer for it?
Quick Reply
Related discussions
- Aerospace/Aeronautical Eng Uni Application
- How do i get into Cambridge natural sciences (physics) from this point on.
- Cambridge chemistry challenge puzzle
- Nat science (physical) without further maths.
- Math AA HL for Biochemistry / Natural Sciences in Oxbridge?
- Can I get into Cambridge or Imperial engineering without doing well in olympiads?
- University Application - Cambridge, Imperial, UCL, Manchester, Eidngurgh
- What should I drop? Chem or AS FM?
- Cambridge Maths School or Hills Road
- I need help for my personal statement
- Am I too dumb to go to Oxbridge?
- Super-curriculars for Oxbridge?
- A Level Options for Medicine
- Oxford gcse grades
- A level subject combination - medicine
- How likely are Cardiff or Liverpool to give me an offer?
- UKChO 2024
- Maths in cambridge
- I can't Improve
- Natural Sciences at Cambridge
Latest
Trending
Last reply 6 days ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 6 days ago
Im confused about this chemistry question, why does it form these products



