AS Chemistry- helping each other out!
Scroll to see replies
I know everything about reducing oxidation redox ect but for the ox. States if the number goes down is it reduced and if number goes up its oxidised
Just want a way that would save me thinking time?
Just want a way that would save me thinking time?
Original post by samwillettsxxx
I know everything about reducing oxidation redox ect but for the ox. States if the number goes down is it reduced and if number goes up its oxidised
Just want a way that would save me thinking time?
Just want a way that would save me thinking time?
Number is reduced, so species is reduced.
Number goes down, species is reduced(or goes down, so to speak)
So for oxidation numbers maybe just remember. Down, Down and Up, Up?
Idk I don't really have a mnemonic for that one to remember it by in any other way.
Original post by samwillettsxxx
I know everything about reducing oxidation redox ect but for the ox. States if the number goes down is it reduced and if number goes up its oxidised
Just want a way that would save me thinking time?
Just want a way that would save me thinking time?
Try OILRIG! oxidation Is Loss Reduction Is Gain! Note, reduction doesn't mean go up though as gaining electrons which are negative pull you down however oxidation is where you give electrons up so positie
(edited 8 years ago)
Original post by thymolphthalein
Number is reduced, so species is reduced.
Number goes down, species is reduced(or goes down, so to speak)
So for oxidation numbers maybe just remember. Down, Down and Up, Up?
Idk I don't really have a mnemonic for that one to remember it by in any other way.
Number goes down, species is reduced(or goes down, so to speak)
So for oxidation numbers maybe just remember. Down, Down and Up, Up?
Idk I don't really have a mnemonic for that one to remember it by in any other way.
Thanks so if it was 0 to -4 it's reduced
-5 to say 2 is oxidised?
Original post by Bhavika22
Try OILRIG! oxidation Is Loss Reduction Is Gain! Note, reduction doesn't mean go up though as gaining electrons which are negative pull you down however oxidation is where you give electrons up so positie
Yeah I do use oil rig I know all that I mean just so I can identify if it's reduced or oxidised quickly by realising if the ox state goes down it's reduced and if the ox state goes up its oxidised
Thank u anyway

Original post by samwillettsxxx
Thanks so if it was 0 to -4 it's reduced
-5 to say 2 is oxidised?
-5 to say 2 is oxidised?
Yes exactly.

I would have said OILRIG (oxidation is loss of electrons, reduction is gain of electrons), but that is more specifically for the loss of electrons.
When you lose electrons, the charge will be postive.
When you gain electrons, the charge will be negative.
But you could have something go from +3 to +5 as well and it is will oxidized, so you can't always depend on it increasing from a negative to postive.
Original post by thymolphthalein
Yes exactly. 
I would have said OILRIG (oxidation is loss of electrons, reduction is gain of electrons), but that is more specifically for the loss of electrons.
When you lose electrons, the charge will be postive.
When you gain electrons, the charge will be negative.
But you could have something go from +3 to +5 as well and it is will oxidized, so you can't always depend on it increasing from a negative to postive.

I would have said OILRIG (oxidation is loss of electrons, reduction is gain of electrons), but that is more specifically for the loss of electrons.
When you lose electrons, the charge will be postive.
When you gain electrons, the charge will be negative.
But you could have something go from +3 to +5 as well and it is will oxidized, so you can't always depend on it increasing from a negative to postive.
Thank u for all the help

I was just wondering if I've named this correctly?

Original post by samwillettsxxx
I was just wondering if I've named this correctly?

Yep.
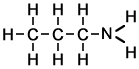

Original post by thymolphthalein
Yep.




Thank u, I know so many questions I'm sorry but in elimination does the OH attack the hydrogen on the carbon to the left of the carbon attached to the halogen
Original post by samwillettsxxx
Thank u, I know so many questions I'm sorry but in elimination does the OH attack the hydrogen on the carbon to the left of the carbon attached to the halogen
Wouldn't that be nucleophilic substituition, if it was a nucleophilic attack?
It would come from the side that the halogen is not on.
Elimination is when halogenoalkanes undergo a reaction with NaOH(or another alkali) to form an alkene, Sodium Halide and water.
Guys please help me, I can't seem to name any thing. I can't name one single thing, I find it so hard to find a pattern with how to name so and so and now I'm going over esterification and esters. Naming things has been a nightmare, people have explained but I still don't get it.
For naming esters, the way I do it is the is somthing-anoate is always the carboxylic acid with the something-anoic acid on the end because both the ends are weird. For the first part which is something-thyl, it will always be whatever else your reacting, for example, if you react propanoic acid and ethanol in the presence of an H2SO4 catalyst, the organic product will be ethyl (since ethanol is the other thing or alcohol) propanoate (because propanoic acid is the carboxylic acid).
I would just like to declare in advanced that I am utterly ****ed for my Chemistry exam this afternoon. Sigh, better start preparing for retakes next year :L
Original post by thymolphthalein
Wouldn't that be nucleophilic substituition, if it was a nucleophilic attack?
It would come from the side that the halogen is not on.
Elimination is when halogenoalkanes undergo a reaction with NaOH(or another alkali) to form an alkene, Sodium Halide and water.
It would come from the side that the halogen is not on.
Elimination is when halogenoalkanes undergo a reaction with NaOH(or another alkali) to form an alkene, Sodium Halide and water.
I didn't mean attack I mean like this sorry can't explain..
when we purify aluminium why do we put ticl4 in an argon atmosphere?
Original post by ashiekh
when we purify aluminium why do we put ticl4 in an argon atmosphere?
To prevent side reactions I think
Edit: just checked to prevent side reactions with oxygen and nitrogen in air

(edited 8 years ago)
Original post by samwillettsxxx
To prevent side reactions I think
Edit: just checked to prevent side reactions with oxygen and nitrogen in air
Edit: just checked to prevent side reactions with oxygen and nitrogen in air

thank youuu
Original post by samwillettsxxx
I didn't mean attack I mean like this sorry can't explain..

Ah yeah, that is elimination. I think your arrows are correct, we were never taught to draw the mechanism for it.
But the equation is correct.
It would form an alkene and Water and Halide ion (from the salt I mentioned earlier)
If there are unsymmetrical haloalkanes, it can form isomeric alkenes because you can shift the double bond.
Original post by thymolphthalein
Ah yeah, that is elimination. I think your arrows are correct, we were never taught to draw the mechanism for it.
But the equation is correct.
It would form an alkene and Water and Halide ion (from the salt I mentioned earlier)
If there are unsymmetrical haloalkanes, it can form isomeric alkenes because you can shift the double bond.
But the equation is correct.
It would form an alkene and Water and Halide ion (from the salt I mentioned earlier)
If there are unsymmetrical haloalkanes, it can form isomeric alkenes because you can shift the double bond.
I do AQA, maybe u do OCR or something that's why you don't have to draw it maybe?
Quick Reply
Related discussions
- TSR Study Together - STEM vs Humanities!
- GCSE Exam Discussions 2024
- Chemistry Degree Lab Work
- Making chemistry notes
- Physical natural sciences
- A level chemistry for a biology degree? (help please!!)
- !!! A level choices - Chem or Computer science
- Do I have enough time to turn my grades around??
- Mass vs relative atomic mass on periodic table
- Official NATURAL SCIENCES applicants thread 2024
- What is it like doing chem vs natural sciences at uni?
- Study leave
- in need of urgent advice, would appreciate any help :)
- Lancaster uni timetable
- Need help on a Venn diagram question
- Biochemistry at University
- Cambridge Natural Sciences Interview Applicants 2023/24
- Higher chemistry
- I enjoy chem practicals more than bio practicals. Is a chem degree right for me?
- Save me from a level torture
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



