Shapes of molecules
Can someone explain how to draw AsF5 and ClF2+.
Like the way I have been taught is to look at the central atom
For the first one As. Count the number of e- in outer shell.
Which is 7 + 5( from the electrons fluorine gives),
12/2 = 6. So square planar?
But why is it trigonal bipyramidal. Like it works with PCl5 but not this?
Like the way I have been taught is to look at the central atom
For the first one As. Count the number of e- in outer shell.
Which is 7 + 5( from the electrons fluorine gives),
12/2 = 6. So square planar?
But why is it trigonal bipyramidal. Like it works with PCl5 but not this?
Basically firstly As has 5 outer shell electrons so draw the atom with the outer shell electrons somewhere on the page, then draw the Fluorine atoms on the As, simple basic covalent bonding diagram. Hence you can see that there are no free electrons on the atom so there are 5 bonding pairs. Let me know if you dont understand.
Original post by Zakria123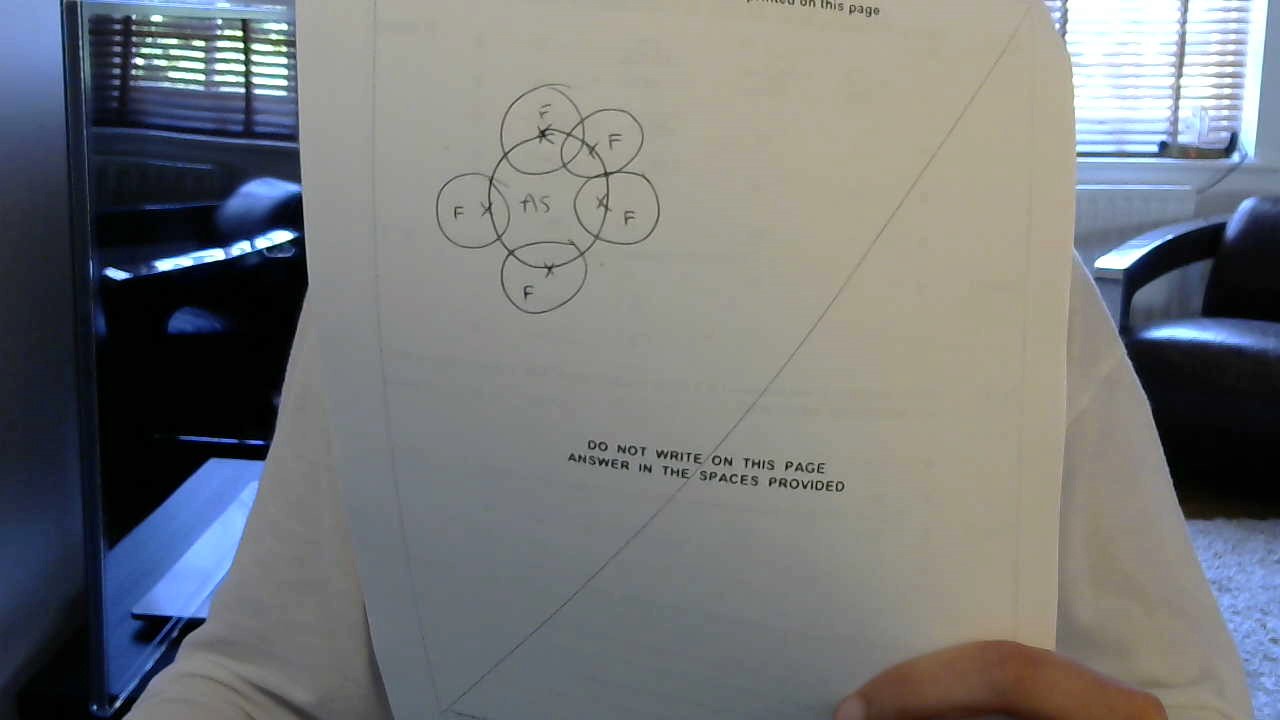
Doesn't As have seven electrons on outer shell? And there are 5 bonded so what happens to the lone pair?
Original post by Super199
Doesn't As have seven electrons on outer shell? And there are 5 bonded so what happens to the lone pair?
OH FFS dw I thought it was At
Original post by Super199
Doesn't As have seven electrons on outer shell? And there are 5 bonded so what happens to the lone pair?
As has 5 outer shell electrons
Original post by Super199
Doesn't As have seven electrons on outer shell? And there are 5 bonded so what happens to the lone pair?
There appears to be a lot of mis-information in this thread.
If the original molecule is AsF5 then you are dealing with arsenic from group 5 (15) NOT astatine from group 7 (17).
There are 5 electrons from the central atom plus one from each fluorine = 10 electrons = 5 electron domains, all of which are used for bonding.
The molecule is trigonal bipyramidal.
Original post by Zakria123
Anyways don't use that method you were using, if they give anything even a tad harder I think you will mess up
With ClF2+ . How do you know there are 2 lone pairs left?
Original post by charco
There appears to be a lot of mis-information in this thread.
If the original molecule is AsF5 then you are dealing with arsenic from group 5 (15) NOT astatine from group 7 (17).
There are 5 electrons from the central atom plus one from each fluorine = 10 electrons = 5 electron domains, all of which are used for bonding.
The molecule is trigonal bipyramidal.
If the original molecule is AsF5 then you are dealing with arsenic from group 5 (15) NOT astatine from group 7 (17).
There are 5 electrons from the central atom plus one from each fluorine = 10 electrons = 5 electron domains, all of which are used for bonding.
The molecule is trigonal bipyramidal.
Yeah I misread it as astatine for some reason lol
Original post by Super199
With ClF2+ . How do you know there are 2 lone pairs left?
Your method is fundamentally correct.
Chlorine = 7 electrons
2 x F = 2 electrons
subtract one for the positive charge
Total electrons = 8 = four pairs, two of which are used for bonding leaving two lone pairs.
Electron domains = 4 = tetrahedral
Ionic shape = bent or angular with two lone pairs.
Original post by charco
Your method is fundamentally correct.
Chlorine = 7 electrons
2 x F = 2 electrons
subtract one for the positive charge
Total electrons = 8 = four pairs, two of which are used for bonding leaving two lone pairs.
Electron domains = 4 = tetrahedral
Ionic shape = bent or angular with two lone pairs.
Chlorine = 7 electrons
2 x F = 2 electrons
subtract one for the positive charge
Total electrons = 8 = four pairs, two of which are used for bonding leaving two lone pairs.
Electron domains = 4 = tetrahedral
Ionic shape = bent or angular with two lone pairs.
Yeah got it cheers

Do you mind helping me with another qs:
An ultrasound imaging agent has the formula C4F10It can be made by the reaction of butane and fluorine as shown in the followingequation.C4H10 + 10F2 →C4F10 + 10HFCalculate the percentage atom economy for the formation of C4F10 in this reaction.Give your answer to three significant figures.
Where does 238 come from?
Original post by Super199
Yeah got it cheers 
Do you mind helping me with another qs:
An ultrasound imaging agent has the formula C4F10It can be made by the reaction of butane and fluorine as shown in the followingequation.C4H10 + 10F2 →C4F10 + 10HFCalculate the percentage atom economy for the formation of C4F10 in this reaction.Give your answer to three significant figures.
Where does 238 come from?

Do you mind helping me with another qs:
An ultrasound imaging agent has the formula C4F10It can be made by the reaction of butane and fluorine as shown in the followingequation.C4H10 + 10F2 →C4F10 + 10HFCalculate the percentage atom economy for the formation of C4F10 in this reaction.Give your answer to three significant figures.
Where does 238 come from?
I can't see a 238 in the question...

Original post by charco
I can't see a 238 in the question... 

In the ms. I dont see where its from?
Original post by Super199
In the ms. I dont see where its from?
... there was no mention of a mark scheme!
238 is the relative mass of C4F10
Original post by charco
Your method is fundamentally correct.
Chlorine = 7 electrons
2 x F = 2 electrons
subtract one for the positive charge
Total electrons = 8 = four pairs, two of which are used for bonding leaving two lone pairs.
Electron domains = 4 = tetrahedral
Ionic shape = bent or angular with two lone pairs.
Chlorine = 7 electrons
2 x F = 2 electrons
subtract one for the positive charge
Total electrons = 8 = four pairs, two of which are used for bonding leaving two lone pairs.
Electron domains = 4 = tetrahedral
Ionic shape = bent or angular with two lone pairs.
Hi,
I use this method myself and works fine but how would you determine the shape if you are given a formula such as NH3? Im unsure how this works since either N or H could be the central element?
Original post by charco
I can't see a 238 in the question... 

In the ms?
In this case N is the central atom with 3 H bonded to it. It would form 3 bonded pairs and 1 lone pair... So it's either trigonal pyramidal/ terahedral depending on what shapes you learnt.
Quick Reply
Related discussions
- shape of molecules a level chem
- Shapes of mocelues
- A level chem dipole question help needed!
- a level chemistry
- a level chemistry drawing moelceules
- multiple choice Q
- Shapes of molecules
- AQA A level chemistry
- a level chemistry
- BSF polarity
- Cycloalkanes
- Chemistry - melting point
- Will the bond angle in H2O(104.5) increase if water is bind to other molecules?
- Biology Paper 2 AQA Triple Higher 2023
- Paper 3 AQA a Level biology
- a level chemistry aqa
- bonding
- NMR- drawing and labelling spectra.
- biology
- how do you identify permanent dipole- permanent dipole molecules?
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



