This discussion is now closed.
Check out other Related discussions
- Any good websites/Youtube Channels for A-Levels?
- TSR Study Together - STEM vs Humanities!
- stem extra curriculars/work experience (year 10)
- Entry requirements help!
- Chemistry at Aston uni
- Specific degree course
- University Application - Cambridge, Imperial, UCL, Manchester, Eidngurgh
- The Official 'I'm going to fail my exams' Society
- How can I plan to manage my time at uni? idk how i will even have time for everything
- AQA vs Edexcel A Level Chemistry and Physics
- Natural Sciences Bath or Durham?
- Uni tips & advice!
- Thinking of going to University of Exeter in September 2024? Ask us a question
- How does one run a medical society????
- What uni for chemistry
- Joining chemical biology society
- Manchester vs Loughborough for Chemistry
- TSR Study Together - STEM vs Humanities - Fifth Session
- Chemistry at Uni.
- Does anyone do Chemical Engineering at Bath?
TSR Chemistry Society
Scroll to see replies
Original post by illusionz
You are correct.
Fine. But there is another question about isotopes: Why it is possible that there are atoms which have more neutrons than protons in a nucleus? in other words: How isotopes come into being?
Original post by D.Bman
Hi illusionz and Ari Ben Canaan, Thank you very much for your help indeed  I really appreciated all the help you have given me
I really appreciated all the help you have given me 
Illusionz can i just have confirmation for Spectra number 1 and number 5 please ?
Especially number 5 because I cant understand from the post weather it is an amine or amide ?
And can you tell me what bonds might be present in this spectra ?
Thank you very much Lads
 I really appreciated all the help you have given me
I really appreciated all the help you have given me 
Illusionz can i just have confirmation for Spectra number 1 and number 5 please ?
Especially number 5 because I cant understand from the post weather it is an amine or amide ?
And can you tell me what bonds might be present in this spectra ?
Thank you very much Lads

I think spectrum 1 is a hydrocarbon without a C=C, O, N etc It is possible there is a halogen but you really can't tell. You would need more data to determine the structure of the compound, NMR and mass spec would be very useful.
As for 5, I do not think it is an amide as an amide C=O is generally ~1680 wavenumbers, and in any case, almost certainly below 1700. The C=O in this case is above 1700 which gives me the impression the C=O and N-H are seperate functional groups and not part of an amide.
Original post by illusionz
I think spectrum 1 is a hydrocarbon without a C=C, O, N etc It is possible there is a halogen but you really can't tell. You would need more data to determine the structure of the compound, NMR and mass spec would be very useful.
As for 5, I do not think it is an amide as an amide C=O is generally ~1680 wavenumbers, and in any case, almost certainly below 1700. The C=O in this case is above 1700 which gives me the impression the C=O and N-H are seperate functional groups and not part of an amide.
As for 5, I do not think it is an amide as an amide C=O is generally ~1680 wavenumbers, and in any case, almost certainly below 1700. The C=O in this case is above 1700 which gives me the impression the C=O and N-H are seperate functional groups and not part of an amide.
Mate for number 1
Can you tell me what would be reason for the hydrocarbon to not have the C=C and O, N having looked at the spectra ?
Is it due to the peaks or something ?
Original post by D.Bman
Mate for number 1
Can you tell me what would be reason for the hydrocarbon to not have the C=C and O, N having looked at the spectra ?
Is it due to the peaks or something ?
Can you tell me what would be reason for the hydrocarbon to not have the C=C and O, N having looked at the spectra ?
Is it due to the peaks or something ?
Because the spectrum doesn't have peaks in the regions these bonds cause peaks to appear.
Hey Everyone 
Can someone help me with a chemistry question
I need to draw just one mechanisms for
Nitration of Benzene
Ethane + Bromine
Ethane + Chlorine
Can someone help me draw these please
Thank you everyone xx
xx

Can someone help me with a chemistry question

I need to draw just one mechanisms for
Nitration of Benzene
Ethane + Bromine
Ethane + Chlorine
Can someone help me draw these please

Thank you everyone
 xx
xxOriginal post by KellyRoth
Hey Everyone 
Can someone help me with a chemistry question
I need to draw just one mechanisms for
Nitration of Benzene
Ethane + Bromine
Ethane + Chlorine
Can someone help me draw these please
Thank you everyone xx
xx

Can someone help me with a chemistry question

I need to draw just one mechanisms for
Nitration of Benzene
Ethane + Bromine
Ethane + Chlorine
Can someone help me draw these please

Thank you everyone
 xx
xxFor the nitration of benzene you are essentially reacting H2SO4 and HNO3 together to produce a Nitronium ion.
Think about the interaction between the two acids.... Can you see a way by which a good leaving group (HINT: water) could be produced on the HNO3 molecule ?
Original post by Ari Ben Canaan
For the nitration of benzene you are essentially reacting H2SO4 and HNO3 together to produce a Nitronium ion.
Think about the interaction between the two acids.... Can you see a way by which a good leaving group (HINT: water) could be produced ?
Think about the interaction between the two acids.... Can you see a way by which a good leaving group (HINT: water) could be produced ?
Right, So im essentially reacting :
H2SO4 + HNO3 ----> Nitronium Ion ( Can you tell me the formula for this
 )
) So after reacting H2SO4 + HNO3 would i expect to see water being produced ?
Can you correct me if am going wrong, because chemistry is my weakest subject

Also what bonding would i see in this mechanism ? xx
Original post by KellyRoth
Right, So im essentially reacting :
H2SO4 + HNO3 ----> Nitronium Ion ( Can you tell me the formula for this )
)
So after reacting H2SO4 + HNO3 would i expect to see water being produced ?
Can you correct me if am going wrong, because chemistry is my weakest subject
Also what bonding would i see in this mechanism ? xx
H2SO4 + HNO3 ----> Nitronium Ion ( Can you tell me the formula for this
 )
) So after reacting H2SO4 + HNO3 would i expect to see water being produced ?
Can you correct me if am going wrong, because chemistry is my weakest subject

Also what bonding would i see in this mechanism ? xx
Are you an A Level student ?
I'm sure you are aware that the electrophile is NO2+ i.e. a nitronium ion.
Essentially, what happens is that the Nitric acid takes a proton from the Sulphuric acid to produce a good leaving group (water).
What is left is the NO2+ ion.
What follows is your usual electrophilic aromatic substitution reaction.
Here, have a look at this :
EDIT : The reaction equation is
HNO3 + 2H2SO4 ---> 2HSO4- + NO2+ + H3O+
(edited 11 years ago)
Original post by Ari Ben Canaan
Are you an A Level student ?
I'm sure you are aware that the electrophile is NO2+ i.e. a nitronium ion.
Essentially, what happens is that the Nitric acid takes a proton from the Sulphuric acid to produce a good leaving group (water).
What is left is the NO2+ ion.
What follows is your usual electrophilic aromatic substitution reaction.
Here, have a look at this :
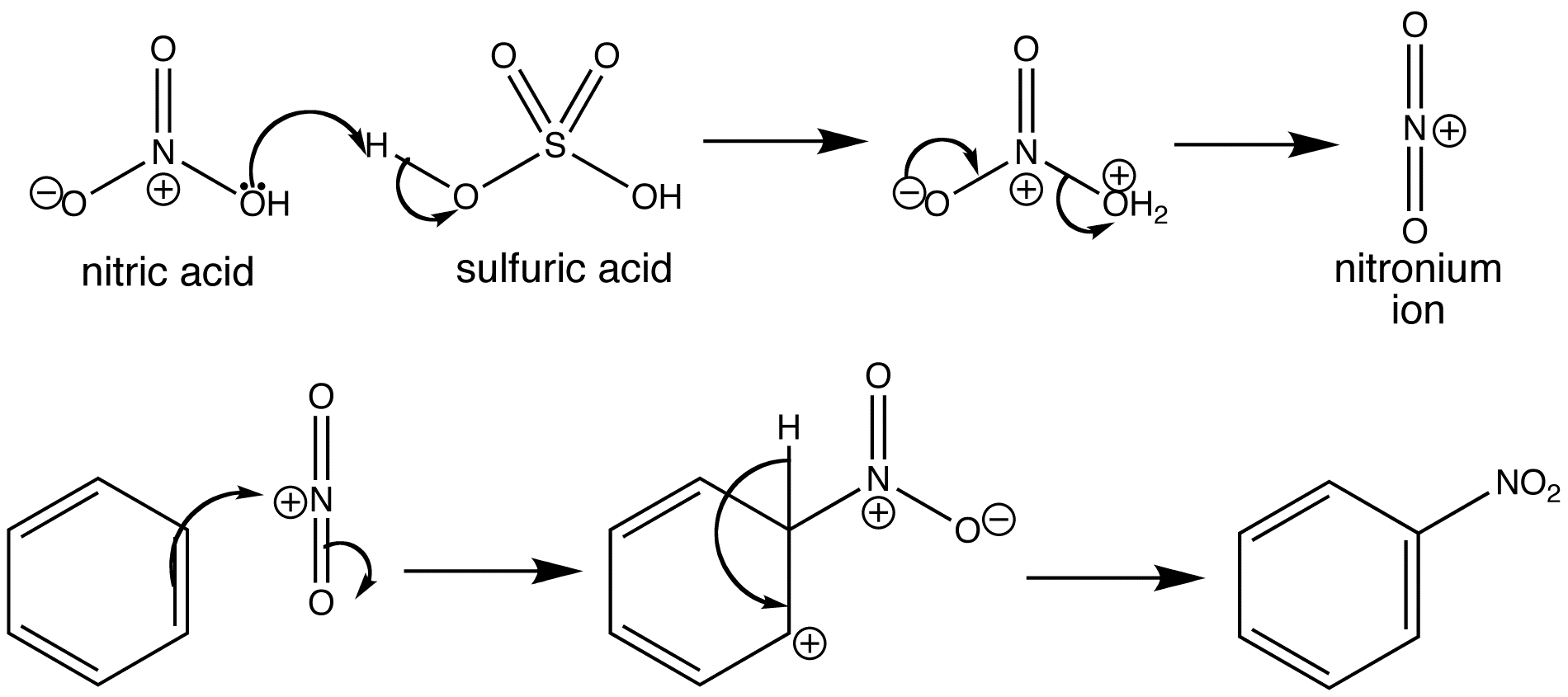
EDIT : The reaction equation is
HNO3 + 2H2SO4 ---> 2HSO4- + NO2+ + H3O+
I'm sure you are aware that the electrophile is NO2+ i.e. a nitronium ion.
Essentially, what happens is that the Nitric acid takes a proton from the Sulphuric acid to produce a good leaving group (water).
What is left is the NO2+ ion.
What follows is your usual electrophilic aromatic substitution reaction.
Here, have a look at this :
EDIT : The reaction equation is
HNO3 + 2H2SO4 ---> 2HSO4- + NO2+ + H3O+
Ah I see, it now makes sense

I understand now that you've added a diagram in, and i can understand whats going on the reaction
And yes i am doing a levels, its just i find chemistry a bit tricky

One thing I am confused about is what type of reaction is it ?
Is it electrophilic aromatic substitution like you have stated above ?
And what type of bonding is going on in this mechanism ?
x
Original post by KellyRoth
Ah I see, it now makes sense 
I understand now that you've added a diagram in, and i can understand whats going on the reaction
And yes i am doing a levels, its just i find chemistry a bit tricky
One thing I am confused about is what type of reaction is it ?
Is it electrophilic aromatic substitution like you have stated above ?
And what type of bonding is going on in this mechanism ?
x

I understand now that you've added a diagram in, and i can understand whats going on the reaction
And yes i am doing a levels, its just i find chemistry a bit tricky

One thing I am confused about is what type of reaction is it ?
Is it electrophilic aromatic substitution like you have stated above ?
And what type of bonding is going on in this mechanism ?
x
The initial interaction between the two acids is a classic acid base reaction. Can you see how the Sulphuric acid donates a proton to the Nitric acid ? We then have a loss of water, which is secondary.
What follows is an electrophilic aromatic substitution.
Type of bonding ? What do you mean ?
Original post by Ari Ben Canaan
The initial interaction between the two acids is a classic acid base reaction. Can you see how the Sulphuric acid donates a proton to the Nitric acid ? We then have a loss of water, which is secondary.
What follows is an electrophilic aromatic substitution.
Type of bonding ? What do you mean ?
What follows is an electrophilic aromatic substitution.
Type of bonding ? What do you mean ?
Oh Right, So we first have an acid base reaction with a proton being donated to the nitric acid. Then a loss of water occours, which is secondary. Then electrophilic subsitution.
I understand it now

Ben Canaan, I was asked in the question to analyse the type of reaction in each case in relation to its bonding.
This is the part where i am confused on, Can you help me please ?
x
I'm back!
I have discussed the fusion between Uranium and Deuterium in the physics society. During the discussion I have read that Uranium and Deuterium applies an energy bonding to become to Plutonium. Why energy come into being during the bonding between Uranium and Deuterium? I suppose that Uranium and Deuterium have to get a stimulated state to come to a bonding, as atoms keep the low-energy state by nature. And when atoms attend a bonding, they are stimulated. That is my supposition.
I have discussed the fusion between Uranium and Deuterium in the physics society. During the discussion I have read that Uranium and Deuterium applies an energy bonding to become to Plutonium. Why energy come into being during the bonding between Uranium and Deuterium? I suppose that Uranium and Deuterium have to get a stimulated state to come to a bonding, as atoms keep the low-energy state by nature. And when atoms attend a bonding, they are stimulated. That is my supposition.
Name: Aleisha Cullen
Hobbies : singing, sport listening to music
studying: AS chemistry, RS, psyschology and English literature
live: westmiddlands
why i like chemo: its logical and interesting
Hobbies : singing, sport listening to music
studying: AS chemistry, RS, psyschology and English literature
live: westmiddlands
why i like chemo: its logical and interesting
please help me with this question !!!
Given that
p(g) + 3Cl(g) = PCl3 (g) energy change = -983kjmol-1
P(s) + 3/2Cl2(g) = PCl3(g) energy change = -305kjmol-1
p(s) = p(g) energy change = +314kjmol-1
Calculate the following bond energies:
a) P-Cl in PCl3
b) Cl-Cl in Cl2
I really have no clue on how to do this so please help :s
Given that
p(g) + 3Cl(g) = PCl3 (g) energy change = -983kjmol-1
P(s) + 3/2Cl2(g) = PCl3(g) energy change = -305kjmol-1
p(s) = p(g) energy change = +314kjmol-1
Calculate the following bond energies:
a) P-Cl in PCl3
b) Cl-Cl in Cl2
I really have no clue on how to do this so please help :s
Original post by Aleisha1
please help me with this question !!!
Given that
p(g) + 3Cl(g) = PCl3 (g) energy change = -983kjmol-1
P(s) + 3/2Cl2(g) = PCl3(g) energy change = -305kjmol-1
p(s) = p(g) energy change = +314kjmol-1
Calculate the following bond energies:
a) P-Cl in PCl3
b) Cl-Cl in Cl2
I really have no clue on how to do this so please help :s
Given that
p(g) + 3Cl(g) = PCl3 (g) energy change = -983kjmol-1
P(s) + 3/2Cl2(g) = PCl3(g) energy change = -305kjmol-1
p(s) = p(g) energy change = +314kjmol-1
Calculate the following bond energies:
a) P-Cl in PCl3
b) Cl-Cl in Cl2
I really have no clue on how to do this so please help :s
Hint : Enthalpy change of reaction = Energy used to Break bonds - Energy used to form bonds.
Original post by Ari Ben Canaan
Hint : Enthalpy change of reaction = Energy used to Break bonds - Energy used to form bonds.
I dont understand why enthalpy change = bonds broken - bonds formed
Posted from TSR Mobile
hi could anyone give me some advice? Basically I am considering being a pharmacist but I don't know that much about it. What different things can a pharmacist do?
Original post by tree123
hi could anyone give me some advice? Basically I am considering being a pharmacist but I don't know that much about it. What different things can a pharmacist do?
Hey! You'd probably be better suited to looking around in and maybe starting a thread in this forum. You're more likely to get an informed reply there.
Original post by Loz17
Hey! You'd probably be better suited to looking around in and maybe starting a thread in this forum. You're more likely to get an informed reply there.
thankyouu! I was trying to find the right place to post it!

Original post by Loz17
Hey! You'd probably be better suited to looking around in and maybe starting a thread in this forum. You're more likely to get an informed reply there.
do you know what jobs I could go into with a chemistry degree?:P
Related discussions
- Any good websites/Youtube Channels for A-Levels?
- TSR Study Together - STEM vs Humanities!
- stem extra curriculars/work experience (year 10)
- Entry requirements help!
- Chemistry at Aston uni
- Specific degree course
- University Application - Cambridge, Imperial, UCL, Manchester, Eidngurgh
- The Official 'I'm going to fail my exams' Society
- How can I plan to manage my time at uni? idk how i will even have time for everything
- AQA vs Edexcel A Level Chemistry and Physics
- Natural Sciences Bath or Durham?
- Uni tips & advice!
- Thinking of going to University of Exeter in September 2024? Ask us a question
- How does one run a medical society????
- What uni for chemistry
- Joining chemical biology society
- Manchester vs Loughborough for Chemistry
- TSR Study Together - STEM vs Humanities - Fifth Session
- Chemistry at Uni.
- Does anyone do Chemical Engineering at Bath?
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products


