free radical reaction confused
In the first propagation step, why is it that the Hydorgen is ALWAYS the one that reacts with the free radical and the spare electron transfers to the other product.
My query is, why specifically does it want to go to the hydrogen, and why is the electron transferred.
My query is, why specifically does it want to go to the hydrogen, and why is the electron transferred.
I’m gonna bump this up for you so someone more capable can answer  I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this day
I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this day
 I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this day
I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this dayOriginal post by Anonymous17!
I’m gonna bump this up for you so someone more capable can answer  I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this day
I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this day
 I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this day
I haven’t looked at anything remotely related to a level chemistry since I finished my course in early May so I’m drawing a blank here. Also, free radical substitution was the one thing I never cracked. Still don’t get it to this dayaww thankss
Original post by vix.xvi
In the first propagation step, why is it that the Hydorgen is ALWAYS the one that reacts with the free radical and the spare electron transfers to the other product.
My query is, why specifically does it want to go to the hydrogen, and why is the electron transferred.
My query is, why specifically does it want to go to the hydrogen, and why is the electron transferred.
Free radicals are VERY reactive. They will react with anything with which they collide.
If chlorine free radicals are formed by homolytic fission stimulated by UV light, then these have no choice but to bump into the hydrogen atoms of the methane, as they surround the carbon atom.
The most likely outcome is HCl and ●CH3
(edited 3 years ago)
Original post by charco
Free radicals are VERY reactive. They will react with anything with which they collide.
If chlorine free radicals are formed by homolytic fission stimulated by UV light, then these have no choice but to bump into the hydrogen atoms of the methane, as they surround the carbon atom.
The most likely outcome is HCl and ●CH3
If chlorine free radicals are formed by homolytic fission stimulated by UV light, then these have no choice but to bump into the hydrogen atoms of the methane, as they surround the carbon atom.
The most likely outcome is HCl and ●CH3
thanks!!
why does the free electron transfer to CH3? Couldn't it just stay with HCl?
Original post by vix.xvi
thanks!!
why does the free electron transfer to CH3? Couldn't it just stay with HCl?
why does the free electron transfer to CH3? Couldn't it just stay with HCl?
You don't have a clear idea of the mechanism. There is no electron transfer involved.
The chlorine free radical collides with the hydrogen on the methyl group, and the electron that was half of the pair holding the hydrogen to the carbon remains on the carbon.
Original post by charco
You don't have a clear idea of the mechanism. There is no electron transfer involved.
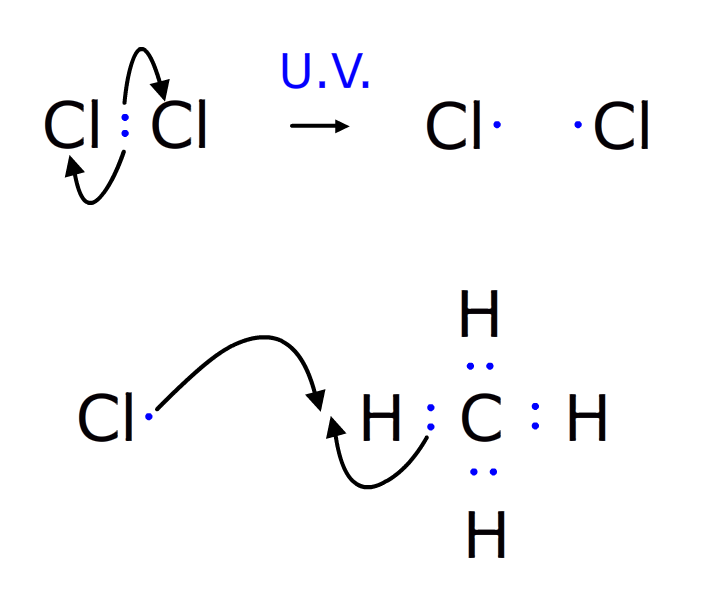
The chlorine free radical collides with the hydrogen on the methyl group, and the electron that was half of the pair holding the hydrogen to the carbon remains on the carbon.
The chlorine free radical collides with the hydrogen on the methyl group, and the electron that was half of the pair holding the hydrogen to the carbon remains on the carbon.
ohhhh
thank you for clearing it up

have a good day
Quick Reply
Related discussions
- Chemistry personal statement.
- very urgent a level chem question
- NO catalysing ozone depletion
- Alkanes MCQ HELP!
- A level chemistry
- Help on this free radical substitution question?
- AlCl3 reacting with benzene
- bio help a level
- Radicals chemistry
- Biochemistry Personal Statement Example
- AQA A Level Chemistry Paper 3 7405/3 - 23 Jun 2022 [Exam Chat]
- Hess law and combustion
- Personal statement help for biological natural sciences
- A level Edexcel Politics
- Rishi Sunak threatens to remove UK from ECHR if Rwanda plan blocked
- le chapeliers principle, exo and endo?????
- Further Mechanics 1 Question
- Nucelophiles Vs Ligands
- A-level Chemistry Study Group 2022-2023
- urgent chemistry question!! a level
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



