Original post by JzANonM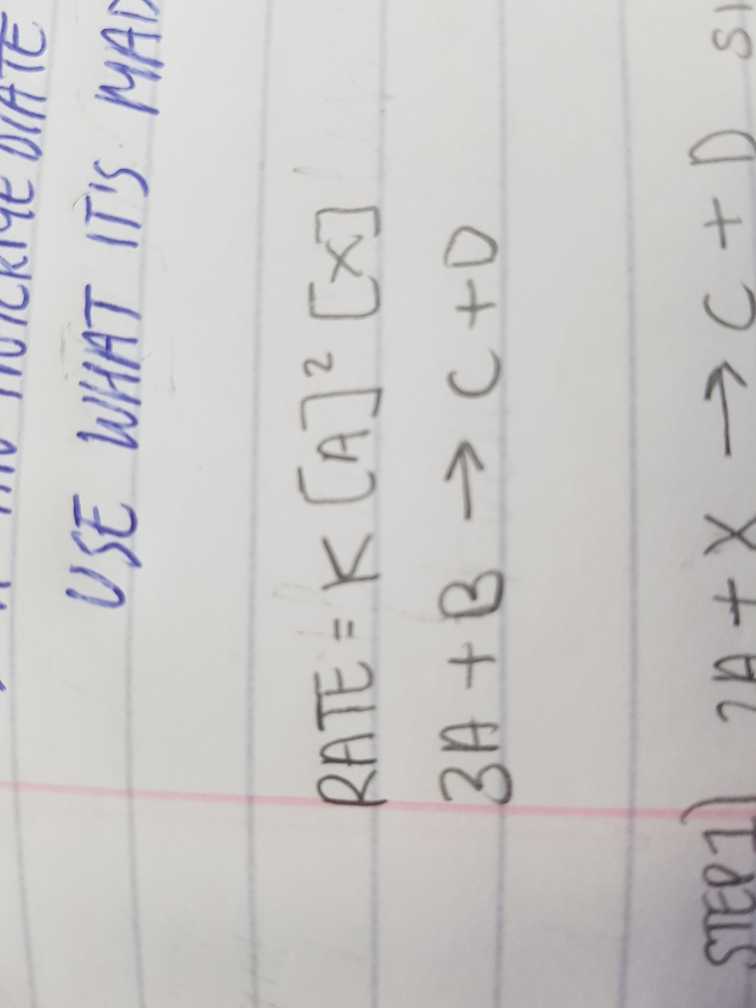
can someone help me, basically my teacher told me to write a 2 step mechanism for this, but im a bit stumped.
can someone help me, basically my teacher told me to write a 2 step mechanism for this, but im a bit stumped.
get smart than.
Original post by masonBrand69
no
great
Original post by masonBrand69
get smart than.
getting smart is acquiring knowledge from others
Original post by p_helena
Your teacher is referring to how you can use the Rate Equation to work out the Rate-Determining Step.
Is (3A + B --> C+ D) the overall equation or one of the steps? this seems poorly explained to me.
Is (3A + B --> C+ D) the overall equation or one of the steps? this seems poorly explained to me.
well yeah the rate determining step is part of the 2 step mechanism
and 3A+B ---> C+D is the overall equation
ahhhh okay cool.
As A is 2nd order (as shown by the ^2), it means that there are 2 moles of A for every 1 mole of everything else in the rate-determining step.
Only substances in the Rate-Determining step are included in the rate equation, so perhaps X is a catalyst that is regenerated later.
E.g. 2A + X ---> C + AX
then B+ A + AX --> D + X
giving 3A + B --> C + D overall.
Hope this kinda makes sense?
As A is 2nd order (as shown by the ^2), it means that there are 2 moles of A for every 1 mole of everything else in the rate-determining step.
Only substances in the Rate-Determining step are included in the rate equation, so perhaps X is a catalyst that is regenerated later.
E.g. 2A + X ---> C + AX
then B+ A + AX --> D + X
giving 3A + B --> C + D overall.
Hope this kinda makes sense?
Original post by JzANonM
well yeah the rate determining step is part of the 2 step mechanism
and 3A+B ---> C+D is the overall equation
and 3A+B ---> C+D is the overall equation
Original post by p_helena
ahhhh okay cool.
As A is 2nd order (as shown by the ^2), it means that there are 2 moles of A for every 1 mole of everything else in the rate-determining step.
Only substances in the Rate-Determining step are included in the rate equation, so perhaps X is a catalyst that is regenerated later.
E.g. 2A + X ---> C + AX
then B+ A + AX --> D + X
giving 3A + B --> C + D overall.
Hope this kinda makes sense?
As A is 2nd order (as shown by the ^2), it means that there are 2 moles of A for every 1 mole of everything else in the rate-determining step.
Only substances in the Rate-Determining step are included in the rate equation, so perhaps X is a catalyst that is regenerated later.
E.g. 2A + X ---> C + AX
then B+ A + AX --> D + X
giving 3A + B --> C + D overall.
Hope this kinda makes sense?
ohhh ok, this makes much more sense than to what I originally thought it was. Thank you!

Not a problem! It's been a while since I did A -Level Chem (about 4 weeks lol) but I think that's the general gist of it.
The main thing to remember is that some substances do not take part in the overall reaction (i.e. are regenerated) but may be in the rate determining step as catalysts.
The only thing I'm unsure on is that catalysts are usually zero order. If what I've said is wrong I'm sorry!
The main thing to remember is that some substances do not take part in the overall reaction (i.e. are regenerated) but may be in the rate determining step as catalysts.
The only thing I'm unsure on is that catalysts are usually zero order. If what I've said is wrong I'm sorry!
Original post by JzANonM
ohhh ok, this makes much more sense than to what I originally thought it was. Thank you! 

Quick Reply
Related discussions
- HELPPPP
- ALEVEL options
- How to go from D to A in a levels
- HELP!!!! Medicine uni!!!!
- Having no active containers may mean that you have no Portico access levels set. Head
- NOTES FOR GCSE URGENT + gimme some tips for grade 9 pls
- Do I have a chance at dentistry? (pleaaassseeeee helpppp)
- Gatsby and Othello comparison question
- criminology unit 4 brief
- yall i got the worst cramps.. ideas to help please?
- Transition metals question-helpppp
- comh and tissue english lit
- Need to grow 2 inches to be a pilot
- About masters program in digital marketing from Uk.
- muslim marriage ?
- Math question
- Helpppp
- How can i get from an E grade to an A* in A-level chemistry???
- Helpppp
- Helpppp
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



