Original post by nour...r
can anyone help me with this objective its in the IAL chemistry syllabus
determine a chemical formula by experiment such as the formula of copper(II) oxide by reduction
if anyone can explain what they mean with an example pls
determine a chemical formula by experiment such as the formula of copper(II) oxide by reduction
if anyone can explain what they mean with an example pls
Also the last three objectives here pls. My external is next week
Original post by nour...r
can anyone help me with this objective its in the IAL chemistry syllabus
determine a chemical formula by experiment such as the formula of copper(II) oxide by reduction
if anyone can explain what they mean with an example pls
determine a chemical formula by experiment such as the formula of copper(II) oxide by reduction
if anyone can explain what they mean with an example pls
I tried explaining it by writing it out but I find it difficult without diagrams so Im going to link a website that seems to have a good method - I would advisements looking at a mark scheme of your exam board to find exactly what you need to write to hit the marks
https://edu.rsc.org/resources/finding-the-formula-of-copperii-oxide/727.article
Original post by gin_n_tonic
I tried explaining it by writing it out but I find it difficult without diagrams so Im going to link a website that seems to have a good method - I would advisements looking at a mark scheme of your exam board to find exactly what you need to write to hit the marks
https://edu.rsc.org/resources/finding-the-formula-of-copperii-oxide/727.article
https://edu.rsc.org/resources/finding-the-formula-of-copperii-oxide/727.article
thank u so much , if u want to share ur writing as well that would be amazing even if its without a diagram. cause my external is coming up and thank u so much again
Original post by gin_n_tonic
I tried explaining it by writing it out but I find it difficult without diagrams so Im going to link a website that seems to have a good method - I would advisements looking at a mark scheme of your exam board to find exactly what you need to write to hit the marks
https://edu.rsc.org/resources/finding-the-formula-of-copperii-oxide/727.article
https://edu.rsc.org/resources/finding-the-formula-of-copperii-oxide/727.article
also I sent an attachment afterwards, if u have an idea about the last three objectives
For the iii practical (with oranges and how to extract Limonene) here's the practical:
(Do not mean to be condescending at all I just can come across that way)
- Place 100cm3 of distilled water (water that has been boiled into its vapour form and then condensed back into liquid but in a different place than where it was originally normal water) into a round bottom flask
- Peel the skin of two oranges by using a grater (the rind)
- Add some small stones which act as anti bumping granules into the flask
- Put this aside and the next thing required is to set up the apparatus which I'm going to link a picture of:
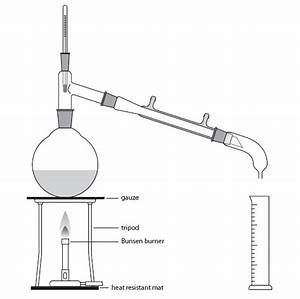 (Is that too small on my screen it seems small, can you click on it or not?)
(Is that too small on my screen it seems small, can you click on it or not?)
- Using the Bunsen burner underneath the flask and reduce the heat/flame accordingly to keep it at more of a simmer as it goes through the process of distillation
- REMEMBER when distillation occurs it is important to keep it at a rate of around 1 drop per second of distillate (make sure it is consistent)
- Using a measuring cylinder, after the adequate amount of time has passed, collect 50cm3 of distillate
- You will see an oil layer on this surface
- Take a pipette (the dropping kind) and transfer this oil layer into a test tube (be very careful doing this as you don't want to lose too much of it)
- Some waft these fumes towards their nose to check for odour but DO NOT DIRECTLY INHALE
- Take three other test tubes and put approximately 1cm3 of bromine water (potassium manganate is usually used in practicals you just have to remember the act of bromine water or to not complicate this just remember bromine water unless you actually have to perform this practical at any point) in each
- Add a few drops of the oil (limonene oil) to ONE of the test tubes
- Add cyclohexane to the second test tube
- Add cyclohexene to the last test tube
- Add the same amount of these chemicals each time (e.g you do two drops in one, make sure you do two drops in all)
- Place in the bungs (like stoppers really)
- Agitate these now to help dissolve some of the chemicals that are harder to break down
- To work out if the molecule contains double bonds, have a look at its appearance and if it has decolourised then that means that it does contain these double bonds
(Do not mean to be condescending at all I just can come across that way)
- Place 100cm3 of distilled water (water that has been boiled into its vapour form and then condensed back into liquid but in a different place than where it was originally normal water) into a round bottom flask
- Peel the skin of two oranges by using a grater (the rind)
- Add some small stones which act as anti bumping granules into the flask
- Put this aside and the next thing required is to set up the apparatus which I'm going to link a picture of:
- Using the Bunsen burner underneath the flask and reduce the heat/flame accordingly to keep it at more of a simmer as it goes through the process of distillation
- REMEMBER when distillation occurs it is important to keep it at a rate of around 1 drop per second of distillate (make sure it is consistent)
- Using a measuring cylinder, after the adequate amount of time has passed, collect 50cm3 of distillate
- You will see an oil layer on this surface
- Take a pipette (the dropping kind) and transfer this oil layer into a test tube (be very careful doing this as you don't want to lose too much of it)
- Some waft these fumes towards their nose to check for odour but DO NOT DIRECTLY INHALE
- Take three other test tubes and put approximately 1cm3 of bromine water (potassium manganate is usually used in practicals you just have to remember the act of bromine water or to not complicate this just remember bromine water unless you actually have to perform this practical at any point) in each
- Add a few drops of the oil (limonene oil) to ONE of the test tubes
- Add cyclohexane to the second test tube
- Add cyclohexene to the last test tube
- Add the same amount of these chemicals each time (e.g you do two drops in one, make sure you do two drops in all)
- Place in the bungs (like stoppers really)
- Agitate these now to help dissolve some of the chemicals that are harder to break down
- To work out if the molecule contains double bonds, have a look at its appearance and if it has decolourised then that means that it does contain these double bonds
Is this the type of thing you want me to do? I can explain why these processes happen if you would like me to as well
Original post by gin_n_tonic
Is this the type of thing you want me to do? I can explain why these processes happen if you would like me to as well
yes pls thank u so much , I really need the help
Original post by gin_n_tonic
I tried explaining it by writing it out but I find it difficult without diagrams so Im going to link a website that seems to have a good method - I would advisements looking at a mark scheme of your exam board to find exactly what you need to write to hit the marks
https://edu.rsc.org/resources/finding-the-formula-of-copperii-oxide/727.article
https://edu.rsc.org/resources/finding-the-formula-of-copperii-oxide/727.article
Good explanation
Do you understand everything so far?
Quick Reply
Related discussions
- Can I do engineering without chemistry in a levels
- Chemistry at Uni?
- Universities for medicinal chemistry
- TSR Study Together - STEM vs Humanities!
- I enjoy chem practicals more than bio practicals. Is a chem degree right for me?
- Chemistry degrees
- Is Chemistry better at A-level than at GCSE?
- A Level subjects for Biological Sciences: advice thread
- Required practicals chemistry A level
- taking a levels I don't like?
- Is there anyway to do extra chemistry outside of school?
- Maths or Chemistry Degree?
- Biomedical Sciences vs Chemistry at Oxford
- What is actually studied in a chemistry degree?
- Crisis of Subjects - I don't know what to study at uni
- Biochemistry undergraduate course
- A-level chemistry revision, taking notes and exam practise help and paper 3
- chemistry at uni?
- Chemistry or Biochemistry?
- Biochemistry at University
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



