Original post by dasda
so with this question, I learn that the hydrogen electrode is standardised as 0 volts. e value of iron -0.44. therefore I think the answer should be Fe|Fe(2+)||(H+)|(h2)|pt. But the answer is Pt|H2|H+
||Fe2+|Fe. I don't get it.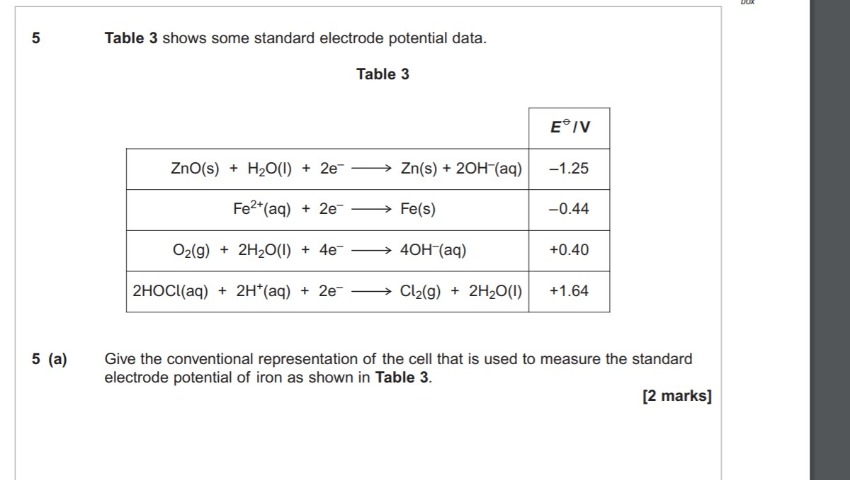
||Fe2+|Fe. I don't get it.
Anode || Cathode
You are correct
Original post by charco
Anode || Cathode
You are correct
You are correct
It has been yonks since it did cell diagrams, but I thought the SHE always goes on the left.
Original post by Pigster
It has been yonks since it did cell diagrams, but I thought the SHE always goes on the left.
I think you are right since that's what the markscheme states.
Original post by Pigster
It has been yonks since it did cell diagrams, but I thought the SHE always goes on the left.
If a reaction contains water and H +, should they be in the cell diagram. I saw that some markschemes leaves out water.
Original post by dasda
If a reaction contains water and H +, should they be in the cell diagram. I saw that some markschemes leaves out water.
The SHE is 2H+ + 2e- <-> H2, i.e. there may be water - so you have H+(aq) - but the water is not part of the reaction.
Original post by Pigster
It has been yonks since it did cell diagrams, but I thought the SHE always goes on the left.
The "convention" is to write cell diagrams as Anode || Cathode.
Maybe it's different for the SHE when it's used to determine the Eº of the half-cell.

This is a new one on me - I'm going to do a little reading.
Original post by charco
The "convention" is to write cell diagrams as Anode || Cathode.
Maybe it's different for the SHE when it's used to determine the Eº of the half-cell.
This is a new one on me - I'm going to do a little reading.
Maybe it's different for the SHE when it's used to determine the Eº of the half-cell.

This is a new one on me - I'm going to do a little reading.
I think on chem guide he talks about how SHE is written on left.
Original post by dasda
I think on chem guide he talks about how SHE is written on left.
Yes, I saw chemguide. The guy who writes it is an A' level examiner, so it's possibly a specification thing.
I can't find any other references and I'm currently separated from the definitive referenced books.
Original post by dasda
I think on chem guide he talks about how SHE is written on left.
OK. It derives from the actual definition of electrode potential. This is from the IUPAC definition:
The so-called electrode potential of an electrode is defined as the emf of a cell in which the electrode on the left is a standard hydrogen electrode and the electrode on the right is the electrode in question. For example, for the silver/silver chloride electrode (written Cl-(aq)|AgCl|Ag) the cell in question is:
Pt|H2(g, p = p°)|HCl(aq)¦HCl(aq)|AgCl|Ag
The cell potential is always measured as the cell is written, so in this case it is +0.20V. The positive sign indicates that the cell reaction is spontaneous as shown and will proceed, with the anode on the left hand side and the cathode on the right hand side.
In summary:
The cell convention shows a spontaneous cell with the anode (oxidation) on the left and the cathode (reduction) on the right. This is spontaneous as written.
If the cell is written the other way round it will not be spontaneous (i.e. the reaction will not happen)
The electrode potential is defined as the cell potential measured when the SHE is on the left hand side of the conventional representation. So if the conventional forward reaction is non-spontaneous, then the reverse reaction will occur and the cell potential, and hence the electrode potential, will be negative as written.
(edited 3 years ago)
Quick Reply
Related discussions
- how do u know when to say ions/electrons
- Electrochemistry
- What is actually studied in a chemistry degree?
- Electrochemistry and effect of pressure
- Chemistry lectures for personal statement
- Electrochemistry help
- chemistry at uni?
- Chem a level - electrochemistry
- AQA A Level Chemistry Electrochemistry
- Chemistry at Uni?
- Urgent help with resistance of water
- AS/A Level Chemistry Study Group 2023/2024
- reuben's y13/medapps journey!!
- Ask a Chemistry Uni Student!
- Holding myself accountable; study!
- AQA A Level Chemistry Paper 3 7405/3 - 23 Jun 2022 [Exam Chat]
- GCSE Exam Discussions 2024
- AQA A-Level Chemistry Paper 3 (7405/3) - 23rd June 2023 [Exam Chat]
- OCR A Level Chemistry Paper 1 H432/01 - 13th Jun 2022 [Exam Chat]
- A Level Maths Checklist
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



