Why does Argon have 18 electrons
Why does argon a nobel gas have 18 electrons in total so 8 in the 3rd shell when it should be 18 electrons
Scroll to see replies
Original post by aspen fernandes
Why does argon a nobel gas have 18 electrons in total so 8 in the 3rd shell when it should be 18 electrons
Like...whatttt??? U r confused!!! Whats ur question?
It has 2 in its innermost shell, 8 in the middle shell, and 8 in the outermost shell, 18 electrons
Original post by aspen fernandes
Why does argon a nobel gas have 18 electrons in total so 8 in the 3rd shell when it should be 18 electrons
Because God wanted it to have 18 electrons.
Spoiler
Original post by Rohit_Rocks10
Like...whatttt??? U r confused!!! Whats ur question?
I am saying nobel gases have a full outer shell but argon doesn't
Original post by aspen fernandes
I am saying nobel gases have a full outer shell but argon doesn't
yes it does, look at my comment above.
s subshell holds 2 electrons
and p holds 6 electrons
it does have a full outer shell.
Argon does have eight electrons in its valence shell. What makes you think that it doesn't?


Original post by aspen fernandes
Why does argon a nobel gas have 18 electrons in total so 8 in the 3rd shell when it should be 18 electrons
It does have 8. Who said it doesnt. 2+8+8=18!!!
Original post by YourGoddamnRight
yes it does, look at my comment above.
s subshell holds 2 electrons
and p holds 6 electrons
s subshell holds 2 electrons
and p holds 6 electrons
well according to A-level Chem textbook and according to Bohr the chemist the 3rd shell has a maximum capacity of 18 electrons but argon doesn't have 18 but 8 electrons.
Original post by aspen fernandes
Why does argon a nobel gas have 18 electrons in total so 8 in the 3rd shell when it should be 18 electrons
The number of total maximum electrons in the nth innermost shell is 2n^2 so the first shell has 2, 2nd has 3, 3rd has 18 etc
In the case of Argon it's configuration goes 2.8.8 so in the 3rd shell it has eight electrons
Bohr's law states the maximum number of electrons that could theoretically be in a shell (the outermost shell doesn't have to be full) but any element has it's own unique number of total electrons that orbit the nucleus and in the case of argon it happens to be 18 electrons. There can't be 18 electrons in the 3rd shell as that would mean there would be 18+8+2 = 28 electrons in total which is obviously false as argon has 18 electrons altogether
(edited 6 years ago)
Original post by Paulington
Argon does have eight electrons in its valence shell. What makes you think that it doesn't?


i am not saying that it doesn't it does have 8 electrons in the outer shell but argon is a nobel gas right so it should have a full outer shell so third shell should contain 18 but argon only has 8
Original post by aspen fernandes
i am not saying that it doesn't it does have 8 electrons in the outer shell but argon is a nobel gas right so it should have a full outer shell so third shell should contain 18 but argon only has 8
But an outer shell can only hold 8 electrons maximum, not 18
Argon has 18 electrons IN TOTAL not just on its outer shell
Original post by m-k1
But an outer shell can only hold 8 electrons maximum, not 18
Argon has 18 electrons IN TOTAL not just on its outer shell
Argon has 18 electrons IN TOTAL not just on its outer shell
if you haven't done AS chem then if you do it then you'll learn that the 3rd shell actually contains 18 electrons, not 8 and the 4th shell contains 32 electrons.
Original post by aspen fernandes
I am saying nobel gases have a full outer shell but argon doesn't
It does
Original post by aspen fernandes
well according to A-level Chem textbook and according to Bohr the chemist the 3rd shell has a maximum capacity of 18 electrons but argon doesn't have 18 but 8 electrons.
I think I understand what you mean:
I'm guessing you are talking about the 3d after the 4s. You're correct that the third shell has a potential for 18 electrons. The 3p^6 (last subshell) fills up the third subshell, if more electrons are added then it would create a 4th shell (4s), then if more electrons are added they would be added to the third shell.
Argon is 1s2, 2s2, 2p6, 3s2, 3p6. The third shell isn't full as it doesn't have any electrons in d-orbitals, but it does have a full p-sub-shell in the 3rd shell so that has the same effect as in previous noble gases. The whole "full outer-shell" thing only really applies to Helium and Neon, all noble gases after them are like Argon in that they have a full p-sub-shell in their outer shell but no electrons in the d-sub-shell. They're still inert, as they are noble gases.
You seem to be confusing the whole "stable octet" thing with electron configuration.
Argon has eight electrons in its outer shell because the third quantum level of orbitals is full. You will learn in A levels that electrons are not as simple as you think, you have multiple orbitals (s, p, d, f, etc) and a "full outer shell" means that the outer (highest energy) quantum level is full.
For example, this is the electron configuration of Argon:
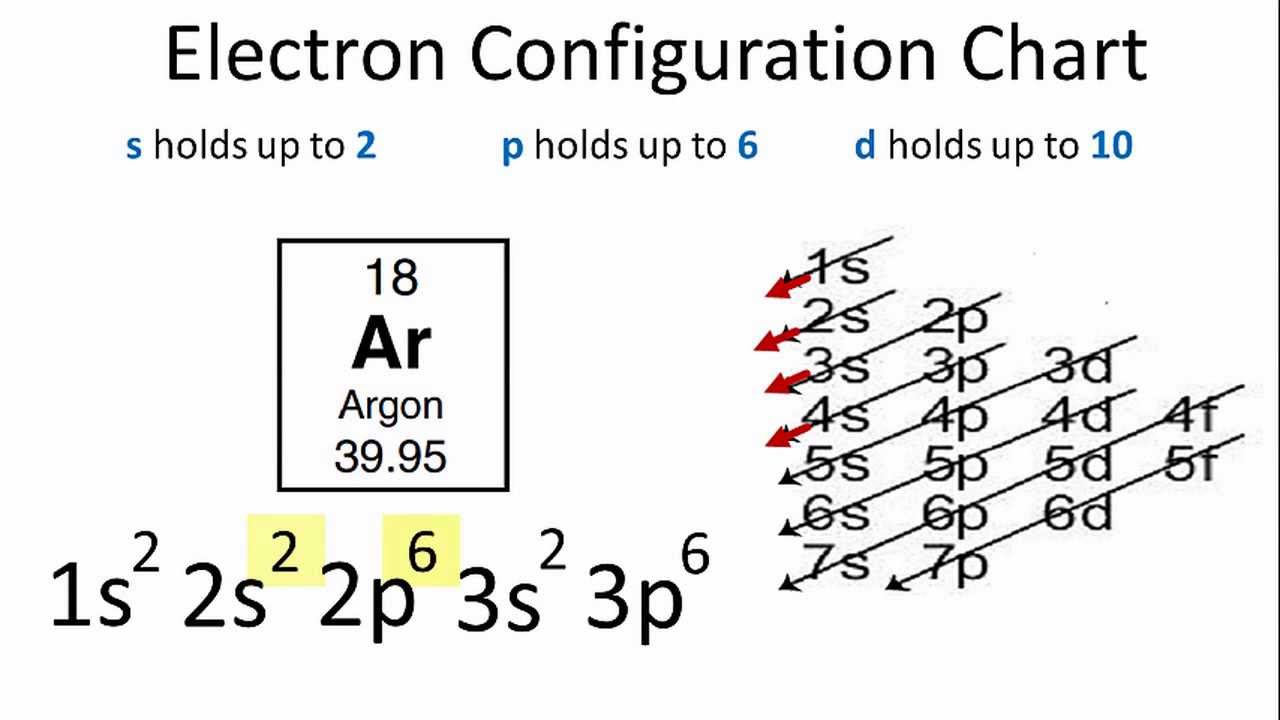
Notice the third quantum level is full (excluding d-orbitals, they're a bit of a special thing) and so it does indeed have a full shell! .
.
Argon has eight electrons in its outer shell because the third quantum level of orbitals is full. You will learn in A levels that electrons are not as simple as you think, you have multiple orbitals (s, p, d, f, etc) and a "full outer shell" means that the outer (highest energy) quantum level is full.
For example, this is the electron configuration of Argon:

Notice the third quantum level is full (excluding d-orbitals, they're a bit of a special thing) and so it does indeed have a full shell!
 .
.In terms of GCSE, outer shell has 2 steps of being full. So it's full at 8 but for potassium and calcium, you have 1 and 2 extra electrons in that shell respectively. Basically it's actually an extremely simplified and inaccurate model that is good at explaining bonding between small atoms.
Quick Reply
Related discussions
- Chemistry A-level exam q help!
- Using your knowledge of chemistry
- Electronic Configurations
- AQA Chem Unit 1 May 22nd 2015 *OFFICIAL THREAD*
- AQA GCSE Chemistry Paper 1 Higher Tier (8462 1H) - 22nd May 2023 [Exam Chat]
- A level question about the 3s orbital
- Pun Help Needed !!
- Chemistry question Orbitals
- chemistry question
- Electrochemistry and effect of pressure
- topics of questions
- A-level Chemistry: Mechanisms
- Why does Nitrogen gain a positive charge in this mechanism ?
- Dissociation chemistry
- (AS AQA Chemistry) Redox reactions - general question
- Chemistry - acids
- What are the storage requirements for 3-nitropropionic acid?
- AQA A level bio help?
- University vs apprenticeship
- Biology - glycolysis
Latest
Trending
Last reply 1 day ago
Edexcel A Level Politics Paper 1 (9PL0 01) - 21st May 2024 [Exam Chat]A-levels
10
Trending
Last reply 1 day ago
Edexcel A Level Politics Paper 1 (9PL0 01) - 21st May 2024 [Exam Chat]A-levels
10




