Which mechanism? & Prediction of products?
Hi guys,
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions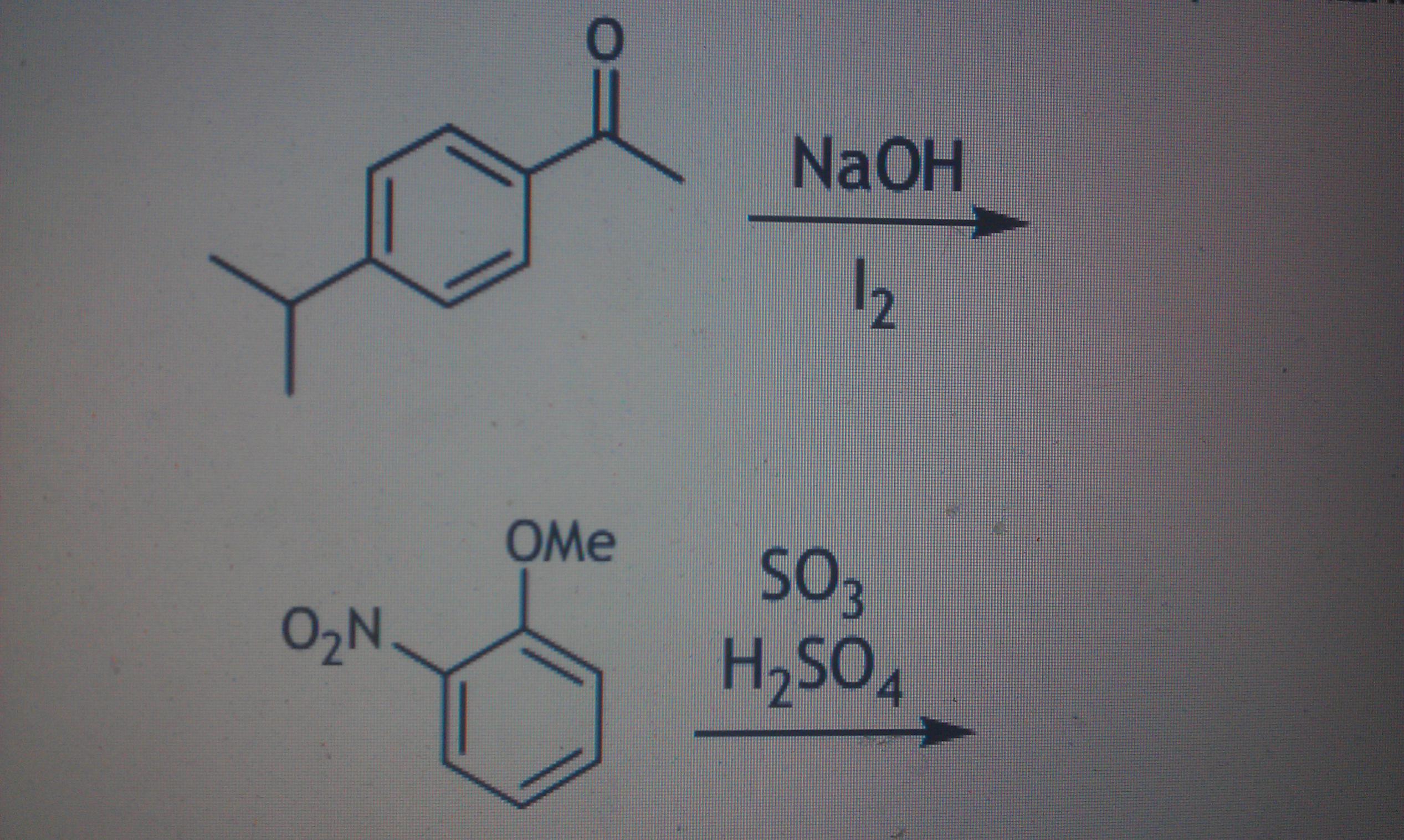
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions
Original post by Princess Kawaii
Hi guys,
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions
I think that you just have to learn the conditions.
The first one is the iodoform reaction. It's a fairly standard test for the CH3CO- group.
The second one looks like the conditions for sulphonation, substitution of an HSO3 group to the ring.
Original post by charco
I think that you just have to learn the conditions.
The first one is the iodoform reaction. It's a fairly standard test for the CH3CO- group.
The second one looks like the conditions for sulphonation, substitution of an HSO3 group to the ring.
The first one is the iodoform reaction. It's a fairly standard test for the CH3CO- group.
The second one looks like the conditions for sulphonation, substitution of an HSO3 group to the ring.
Original post by Princess Kawaii
Hi guys,
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions
Yeah second that. You need to think about sterics for the position of sulphonation.
Original post by Princess Kawaii
Hi guys,
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions
Having a bit of trouble...but how can you tell which mechanism the question wants when it gives you just the reactants and solvent? Also how do you what is going to react with what to get the right product?
Does any one have tips/ steps on how tackle such questions?
E.g these two questions
The first reaction is the iodoform reaction. This reaction occurs with methyl ketones or alcohols which when oxidised give methyl ketones. In this reaction, the methyl ketone group on the ring will be converted into that of methanoic acid.
The second reaction is sulfonation. You have to rationalise where the pi electrons will attack the S03H+ ion. OMe is o/p directing and NO2 is meta directing, so where both those positions coincide is the para position, hence directly opposite to the OMe substituent.
(edited 10 years ago)
Original post by LeonVII
The first reaction is the iodoform reaction. This reaction occurs with methyl ketones or alcohols which when oxidised give methyl ketones. In this reaction, the methyl ketone group on the ring will be converted into that of methanoic acid.
The second reaction is sulfonation. You have to rationalise where the pi electrons will attack the S03H+ ion. OMe is o/p directing and NO2 is meta directing, so where both those positions coincide is the para position, hence directly opposite to the OMe substituent.
The second reaction is sulfonation. You have to rationalise where the pi electrons will attack the S03H+ ion. OMe is o/p directing and NO2 is meta directing, so where both those positions coincide is the para position, hence directly opposite to the OMe substituent.
Well there are 2 positions if you justify it on those grounds... That explanation is true but as I said you need to account for steric effects which leads to the selectivity over the position para to the oxygen.
Posted from TSR Mobile
Original post by JMaydom
Well there are 2 positions if you justify it on those grounds... That explanation is true but as I said you need to account for steric effects which leads to the selectivity over the position para to the oxygen.
Posted from TSR Mobile
Posted from TSR Mobile
Of course, but the steric interactions would make it extremely unfavourable, I thought maybe that was too obvious to post. Maybe i should be a bit more thorough

Original post by LeonVII
Of course, but the steric interactions would make it extremely unfavourable, I thought maybe that was too obvious to post. Maybe i should be a bit more thorough 

Unfavourable yes, but I think you would still get a mixture.
(edited 10 years ago)
Original post by illusionz
You would still get a mixture, despite the sterics.
Indeed, but the percentage of the 1,2 product would be dwarfed by the other, no?
Original post by LeonVII
Indeed, but the percentage of the 1,2 product would be dwarfed by the other, no?
Minor product indeed but any exam answer should, in my opinion, state that both are formed as it gives a more detailed description of the reaction and shows you understand what is going on.
Quick Reply
Related discussions
- Units help
- For each of the following reactions, predict the structures of the main products that
- Ask Us Anything!
- Studying Mechanical Engineering after Product Design
- I don't know what subject to pick
- Help choose last choice uni
- Advanced Higher Mechanics
- Y12 end of years and Y13 predictions
- 2023 Engineering applicants
- Ask a BU Student Rep
- Aero-mechanical engineering???
- 2024 Engineering Applicants
- 2022 Aeronautical and Aerospace Engineering applicants
- Integrated mechanical and electrical engineering vs pure mechanical
- Could someone please help with a mechansim question.
- ce's gygs & stem blog <3
- What can I do to make up for no further maths?
- Official Aston University 2023 Applicant Thread
- A Level Chemistry Organic Help
- Quality/Manufacturing Eng vs Mech Eng
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



