Organic Chemistry
https://www.exam-mate.com//questions/17434/52675/17434_q_52675_38_1.pngCan you please help me with this question. Thank you
The link doesn’t seem to be working could you send a screenshot 

Original post by Blabberboy
https://www.exam-mate.com//questions/17434/52675/17434_q_52675_38_1.pngCan you please help me with this question. Thank you
The page is not found. Maybe you can ask the question here at this place. That would be nice.
Original post by Kallisto
The page is not found. Maybe you can ask the question here at this place. That would be nice.
https://www.exam-mate.com//questions/17434/52675/17434_q_52675_38_1.png
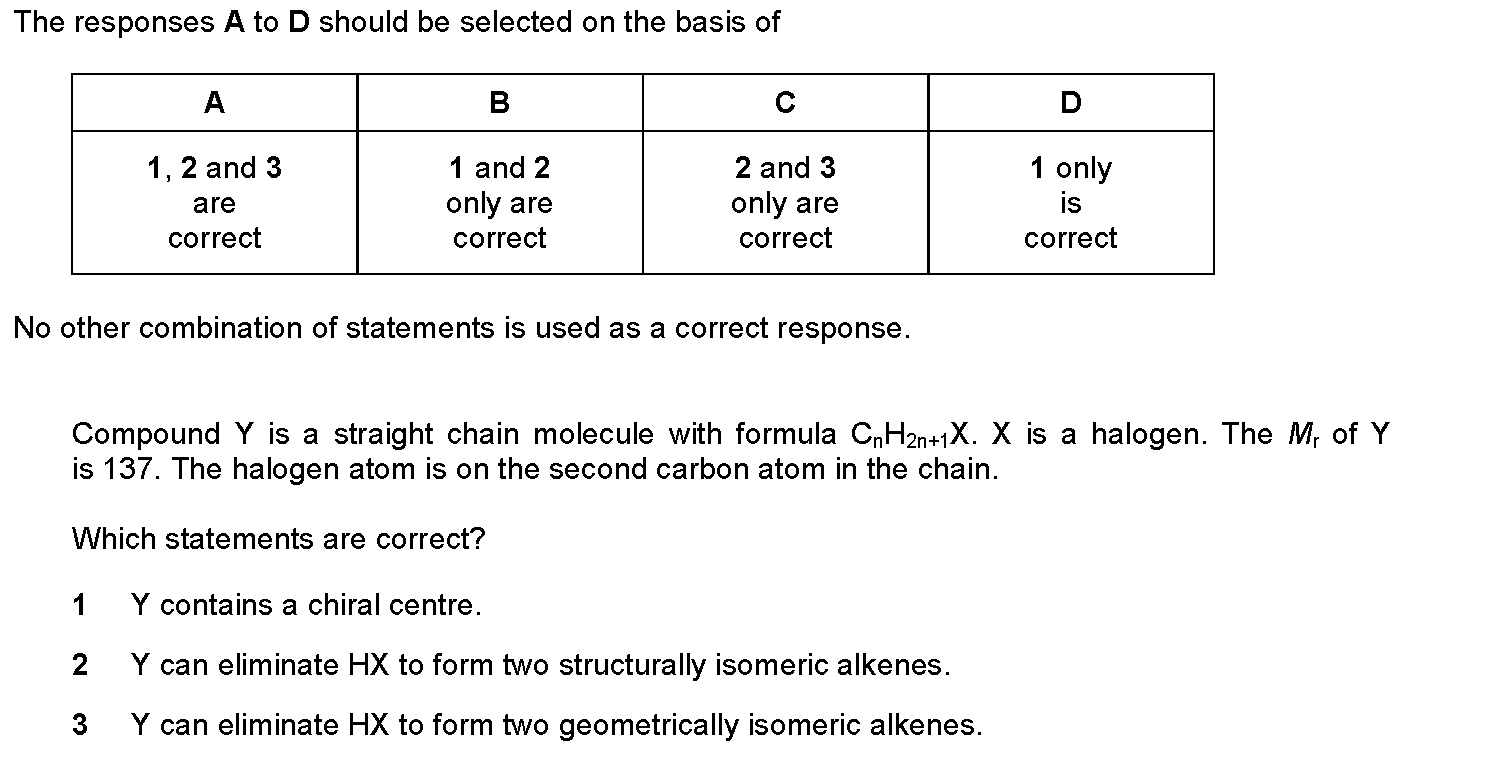
Is this it?
yes plz
Well you don’t know the Mr of the halogen but you can form an equation for the Mr of CnH2n+1
Use multiples of this and try and find one which can be added to a halogens Ar to equal 137
You can then draw out the molecule
Use multiples of this and try and find one which can be added to a halogens Ar to equal 137
You can then draw out the molecule
You can rule out Cl being the halogen as the Mr doesn't end in .5 too 

The answer is 1 2 and 3, all are correct, but i need reasons. I understand 1, but not 2 and 3
What’s n? And X
Don’t worry I think I’ve got it CH3CHBrCH2CH3
Don’t worry I think I’ve got it CH3CHBrCH2CH3
(edited 3 years ago)
Do you understand what structural isomers are and what geometric isomers are?
What’s needed for a geometric isomerism specifically E-Z isomers?
What’s needed for a geometric isomerism specifically E-Z isomers?
Original post by Blabberboy
The answer is 1 2 and 3, all are correct, but i need reasons. I understand 1, but not 2 and 3
As X stands for a halogen, Cl for instance, so HX is HCl. This type of reaction is Dehydrohalogenation: one H- and the Cl-Atom in the halogenalkane are ruptured, formed to HCl and an alkene is left.
You might also want to think about the mechanism for elimination - what do you know about where the hydroxide ion attacks?
Spoiler
Original post by Blabberboy
The answer is 1 2 and 3, all are correct, but i need reasons. I understand 1, but not 2 and 3
Or is it about the isomers? structural isomers in alkenes exists, if the double bound is on a different C-atom, geometric isomer in turn, if the alkene has CH3-branches on a different C-atom (E-Z-isomers!).
(edited 3 years ago)
Quick Reply
Related discussions
- Organic chemistry textbook (uni)
- aqa a level chemistry paper 2 assessed topics
- Chemistry at Uni?
- physical or inorganic??
- How can I become obsessed with chemistry ??
- Chemistry revision
- A level chemistry for a biology degree? (help please!!)
- Tips for physical A level chemistry OCR A?
- AS/A Level Chemistry Study Group 2023/2024
- UCL Natural Sciences and streams
- Is Chemistry better at A-level than at GCSE?
- How should I study organic chemistry?
- Biochem to Med chem
- Inorganic Chemistry (AQA) - A level
- Learning techniques
- Chemistry or Biochemistry?
- AQA A-Level Chemistry Paper 2 (7405/2) - 18th June 2024 [Exam Chat]
- OCR A Chemistry Predictions
- Bath Mpharm Interview Questions
- I enjoy chem practicals more than bio practicals. Is a chem degree right for me?
Latest
Trending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these productsTrending
Last reply 1 week ago
Im confused about this chemistry question, why does it form these products



